Eagle Biosciences is excited to bring you a wide array of more complement assays!
The complement system is an essential part of the immune system in the human body, playing a crucial role in defending against infections, clearing cellular debris, and promoting inflammation. It consists of a complex network of proteins and molecules that work together to enhance the immune response. The complement system can be activated through three main pathways, the classical pathway, the alternative pathway, the lectin pathway. The complement system is tightly regulated to prevent excessive immune responses and potential damage to host tissues. Various regulatory proteins are involved in controlling the activation and amplification of the complement cascade to maintain a delicate balance between defense and protection.
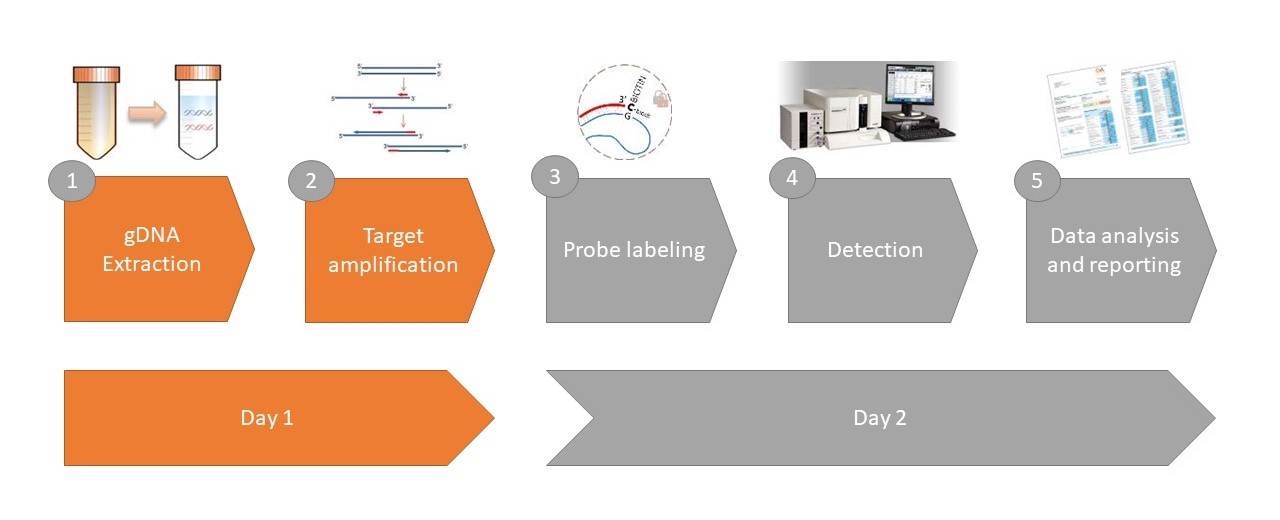
New Complement Assays
Rat Classical Complement Pathway ELISA Assay Kit
Rat Lectin Complement Pathway ELISA Assay Kit
Rat Alternative Complement Pathway ELISA Assay Kit
Mouse Classical Complement Pathway ELISA Assay Kit
Mouse Alternative Complement Pathway ELISA Assay Kit
Pig Classical Complement Pathway ELISA Assay Kit
Pig Lectin Complement Pathway ELISA Assay Kit
Pig Alternative Complement Pathway ELISA Assay Kit
Pig Complement Pathway ELISA Assay Kits
MASP1/C1-INH Complex ELISA Assay Kit
C3d ELISA Assay Kit
MASP-2 ELISA Assay Kit
Complement Factor H ELISA Assay Kit
Complement Factor D ELISA Assay Kit
Complement Factor I ELISA Assay Kit
Collectin-10 ELISA Assay Kit
Factor B ELISA Assay Kit
sCD59 ELISA Assay Kit
C1s/C1-INH Complex ELISA Assay Kit
Rat Terminal Complement Complex (TCC) ELISA Assay Kit
Mouse C3 ELISA Assay Kit
Mouse C1q ELISA Assay Kit
Mouse C3b ELISA Assay Kit
Mouse C4 ELISA Assay Kit
Find the complete complement product catalog here. If you have any questions about any of these products or our other offerings, contact us here.