Angiopoietin-2 ELISA Assay Kit
The Angiopoietin-2 ELISA Assay Kit is Developed and Manufactured in Austria by Biomedica
Size: 1×96 wells
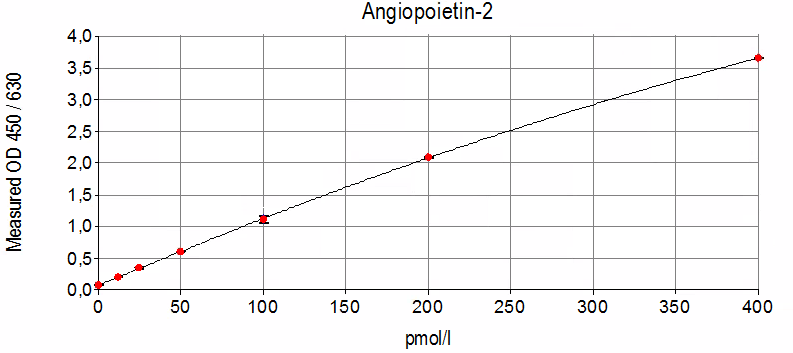
Sensitivity: 3.7 pmol/l
Dynamic Range: 0 – 400 pmol/l
Incubation Time: 3.5 hours
Sample Type: Serum, EDTA plasma, citrate plasma, heparin plasma
Sample Size: 20 µL
Alternative Name: ANG2 ELISA
For Research Use Only
Controls Included
Assay Background
Angiopoietin-2 (ANG2) is a 56.9 kDa glycosylated growth factor that is specific for endothelial cells (ECs). ANG2 is expressed in embryonic vessels and contributes to the formation of new vasculature. In adults, it is restricted to sites of vascular remodeling (e.g. ovary, uterus, placenta) and wound healing. ANG2 is regulated by the cytokine vascular endothelial growth factor (VEGF). Together with VEGF, ANG2 induces endothelial cell migration, proliferation, and vascular sprouting. During angiogenesis, ANG2 exerts its effects via the angiopoietin-1/TIE2 receptor signaling system on endothelial cells. Disruption of this signaling leads to the loss of endothelial integrity. In consequence, the endothelium responds to various pro-inflammatory cytokines and growth factors. Thus, ANG2 might cause vascular micro-inflammation in patients with chronic kidney disease (CKD). Various studies demonstrated that ANG2 levels increase with CKD stage and are associated with fluid overload and abnormal cardiac structure. Furthermore, ANG2 concentrations correlate with mortality in patients with CKD stages 4–5. Although ANG2 levels recover after successful kidney transplantation, ANG2 continues to be a cardiovascular risk factor in this population. In cancer, targeting the TIE2-Angiopoietin pathway has shown promising results in some pre-clinical and clinical trials, including studies on recurrent or metastatic breast and renal cell carcinomas.
Areas of interest:
- Ischemic pathologies (PAD, CAD)
- Inflammation (Bowel disease, Chron’s disease, cirrhosis, sepsis)
- Autoimmune disease (rheumatoid arthritis, psoriasis)
- Artherosclerosis
- Chronic kidney disease
- Diabetic retinopathy
- Cancer
Related Products to Angiopoietin-2 ELISA
Big Endothelin-1 ELISA Assay
Endostatin ELISA Kit
Sclerostin ELISA