Chemerin ELISA Assay Kit
The Chemerin ELISA Assay Kit is For Research Use Only
Size: 1×96 wells
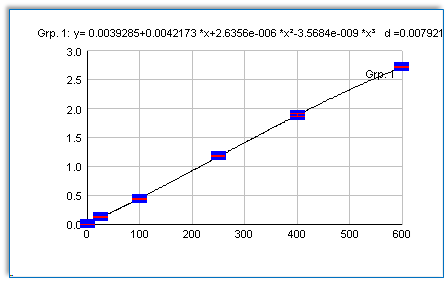
Sensitivity: 5 pg/ml
Incubation Time: 2.5 hours
Sample Type: Serum, Plasma
Sample Size: 10 µL
Alternative Names: TIG2, RARRES2
Product manufactured by Mediagnost
Assay Background
Chemerin, also known as tazarotene-induced gene 2 (TIG2) or retinoic acid receptor responder 2 (RARRES2), is synthesized as precursor protein of 163 amino acids including a N-terminal signal peptide of 20 amino acids, which is chipped of enduring secretion. The inactive, circulation proform of Chemerin contains six cystein residues and thus three intramolecular disulfide bridges are suggested.
Prochemerin expression has been demonstrated for liver, lung, pituitary, lymph node, stomach and adipose tissue (1, 2). It has been detected in blood, ascitic fluids from ovary and liver cancer and synovial fluids from arthritic patients. Different receptors have been found in spleen, lymph node, small intestine, lung tissue as well as in macrophages and immature dendritic cells.
Prochemerin is converted into its biologically active form by serine or cystein proteases, resulting in pro- or anti-inflammatory actions of the active protein, respectively. Proteolytic cleavage of Chemerin has been described for following serine proteases tryptase, plasmin, elastase, cathepsin G as well as for the cross-reactivity proteases cathepsin S and calpains.
The active protein is involved in innate and adaptive immune responses and for instance acts as a strong chemoattractant for immature dendritic cells and macrophages. It influences intracellular signaling by binding to its specific receptors. The C-terminal domain of Chemerin allows binding to the receptor ChemR23/CMKLR1 and elicits a pro-inflammatory stimulus by inducing Ca2+ influx and ERK1/2 activation. Further, the N-terminal domain of Chemerin binds to the CCRL2 receptor and allows presentation of the C-terminal domain to neighbor cells. In case of Prochemerin cleavage by cystein proteases the resulting peptides act inhibitory on the ChemR23/CMKLR1 receptors and thus have anti-inflammatory effects.
Recently, the relevance of Chemerin in adipogenesis and adipocyte metabolism has been discovered. Goralski et al. demonstrated in mice that Chemerin as well as receptor (ChemR23) expression is present in adipocytes of visceral and subcutaneous adipose tissue. The expression and secretion increases enduring adipocyte differentiation. The active Chemerin of 16 kDa has been found in the conditioned media of adipocytes. Intracellular signaling networks in adipocytes are also influenced by Chemerin e.g. stimulation of MAPK p42/44 (ERK1/2) phosphorylation was demonstrated. Further, the authors also show a post-differentiational effect of Chemerin on gene expression in adipocytes.
Based on these results an influence of Chemerin on metabolic syndrome and insulin resistance was purposed. This studies reveal that Chemerin is more expressed by adipose tissue of obese humans and is able to impair insulin sensitivity of muscle cells and insulin mediated lipolysis and lipogenesis in adipocytes. It has been shown that Chemerin is associated with markers of inflammation and components of the metabolic syndrome as well as with renal function.
For the further investigation of Chemerin functions and its quality as biomarker in metabolic diseases reliable measurement of Chemerin in different body fluids is a prerequisite. The Eagle Biosciences Chemerin ELISA Assay Kit offers a sensitive and reproducible test system for the quantitative measurement of Chemerin. Based on highly specific antibodies total Chemerin is measured.