Activated Carboxylated Osteocalcin ELISA
Activated Carboxylated Osteocalcin ELISA Developed and Manufactured in the USA
Size: 1×96 wells
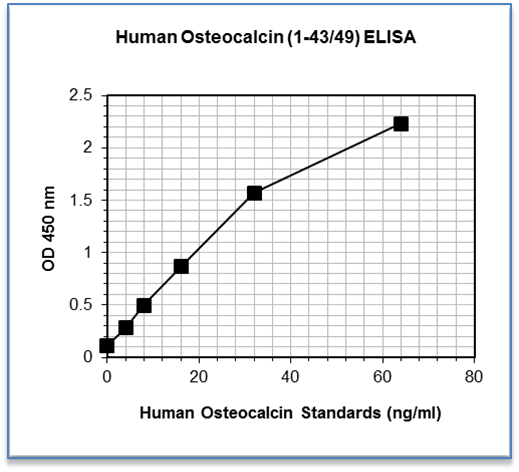
Sensitivity: 0.31 ng/mL
Dynamic Range: 4.0 – 64 ng/mL
Incubation Time: 1.5 hours
Sample Type: Serum, Plasma
Sample Size: 25 µL
Alternative Names: Human Activated Carboxylated Osteocalcin ELISA, Human cOC ELISA, Osteocalcin 1-43/49
For Research Use Only
Controls Included
Assay Principle
The Activated Carboxylated Osteocalcin ELISA Kit is designed, developed and produced for the quantitative measurement of human osteocalcin (1-49) and (1-43) in serum or plasma sample. The assay utilizes the two-site “sandwich” technique with two selected antibodies that bind to different epitopes of human osteocalcin.
Assay standards, controls and samples are added directly to wells of a microtiter plate that is coated with streptavidin. Subsequently, a mixture of biotinylated human osteocalcin N-terminal region specific polyclonal antibody and a peroxidase labeled human osteocalcin 20 – 43 region specific monoclonal antibody is added to each well. After the first incubation period, a “sandwich” of “biotinylated antibody – human osteocalcin – HRP-monoclonal antibody” is formed and this immunocomplex is also captured to the wall of microtiter plate via a streptavidin-biotin affinity binding. The unbound monoclonal antibodies and buffer matrix are removed in the subsequent washing step. A substrate solution in a timed reaction is then measured in a spectrophotometric microplate reader. The enzymatic activity of the immunocomplex bound to the wall of each microtiter well is directly proportional to the amount of human osteocalcin in a test sample. A standard curve is generated by plotting the absorbance versus the respective human osteocalcin concentration for each standard on point-to-point or 4 parameter curve fit. The concentration of human osteocalcin in test samples is determined directly from this standard curve.
Products Related to Activated Carboxylated Osteocalcin ELISA
Anti-CaSR IgG ELISA Assay Kit
Osteonectin (ON) ELISA Assay Kit
Osteoprotegerin (OPG) ELISA Assay Kit