Total FGF-21 ELISA Assay
Total FGF21 ELISA Assay Developed and Manufactured in the USA
Size: 1×96 wells
Sensitivity: 10.4 pg/mL
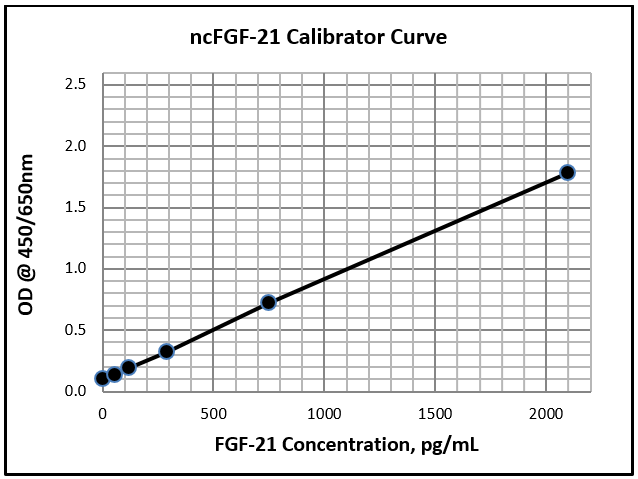
Dynamic Range: 54.0 – 2100 pg/ml
Incubation Time: 2.5 hours
Sample Type: Serum, Plasma
Sample Size: 100 µL
Alternative Names: Fibroblast Growth Factor 21, FGF21
For Research Use Only
Controls Included
Assay Background of Total FGF-21 ELISA
Fibroblast Growth Factor 21 (FGF-21) belongs to the FGF-19 subfamily, which includes FGF-19, FGF-21 and FGF-23. The FGF-19 family members are potent endocrine hormones in the regulation of a diverse physiological homeostasis. The intact FGF-21 is a small protein comprising 181 amino acids. Administration of recombinant FGF-21 lowered plasma glucose and insulin levels, reduced hepatic and circulating triglycerides and cholesterol levels, and improved insulin sensitivity, energy expenditure, hepatic steatosis and obesity in a range of insulin-resistant animal models. The physiological functions of FGF-21 are relied on the intact molecular structure and amino acid sequence in its N-terminal and C-terminal region. The C-terminal nontruncated FGF-21 is a potent cell membrane β-Klotho binder. Whereas, a C-terminal truncated FGF-21 (1-170) is a potent inhibitor that competitively inhibits the biological activity of intact FGF-21 (1-181). Therefore, it is important to measure the circulation intact FGF-21 level in the assessment of the physiological and pathophysiological condition. An assay that determines the fragment of the FGF-21 might overestimate the biological activity of the protein in test sample.
Circulation FGF-21 is a biomarker and its levels are increased in patients with nonalcoholic fatty liver disease (NAFLD), type 2 diabetes, gestational diabetes and obesity. An increase of circulating FGF-21 is also found in patients with Cushing’s syndrome, patients with lipodystrophy induced by HIV-1 and patients with chronic renal disease or end-stage renal disease (ESRD).
Additional FGF-21 Assays Available
C-Terminal FGF-21 ELISA Assay Kit
N-Terminal FGF-21 ELISA Assay Kit
Intact FGF-21 ELISA Assay Kit