Lispro NL-ELISA Assay Kit
The Lispro NL-ELISA Assay Kit was manufactured in Sweden by Mercodia
Size: 12 x 8 wells
Sensitivity: 0.36 mU/L
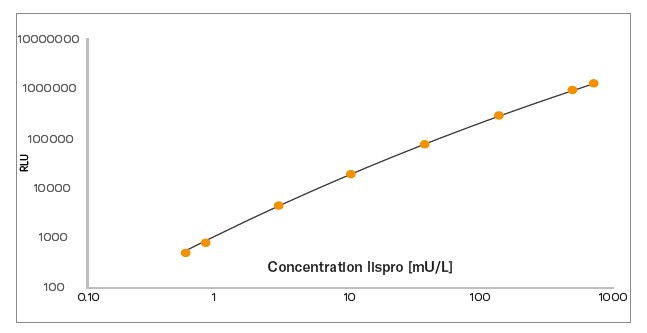
Dynamic Range: 1 – 500 mU/L
Incubation Time: 2 hour 10 minutes
Sample Type: human and porcine serum, EDTA plasma and P-800 plasma
Sample Size: 10 µL
Assay Background
The Lispro NL-ELISA is a ligand binding assay able to measure insulin lispro specifically, without cross-reaction to native insulin, native proinsulin, or any of the tested insulin analogues. Also, no interference from insulin autoantibodies (IgG antibodies), which can be present in samples from diabetic patients, has been observed1. This kit is designed to fit the needs of the pharmaceutical industry, by meeting the requirements specified in the EMA2/FDA3 guidelines. Seven serum calibrators (incl. Calibrator 0), two anchor points and three controls are included in the kit.
Assay Principle
The Lispro NL-ELISA Assay Kit is a solid phase two-site enzyme immunoassay based on the sandwich technique, in which two monoclonal antibodies are directed against separate antigenic determinants on the lispro molecule. Insulin lispro in the sample reacts with anti-lispro antibodies bound to microtitration wells and peroxidase-conjugated anti-lispro antibodies in the solution. A simple washing step removes unbound enzyme-labelled antibody. The bound conjugate is detected by reaction with the chemiluminescent substrate. A chemiluminescence plate reader is used to read the intensity of light generated.
Related Products
Mouse/Rat Proinsulin ELISA Assay Kit
Proinsulin ELISA Assay Kit
Insulin ELISA Assay Kit