FluoBolt Asporin Fluorescence Immunoassay
The FluoBolt Asporin Fluorescence Immunoassay is manufactured in Austria by Fianostics GmbH
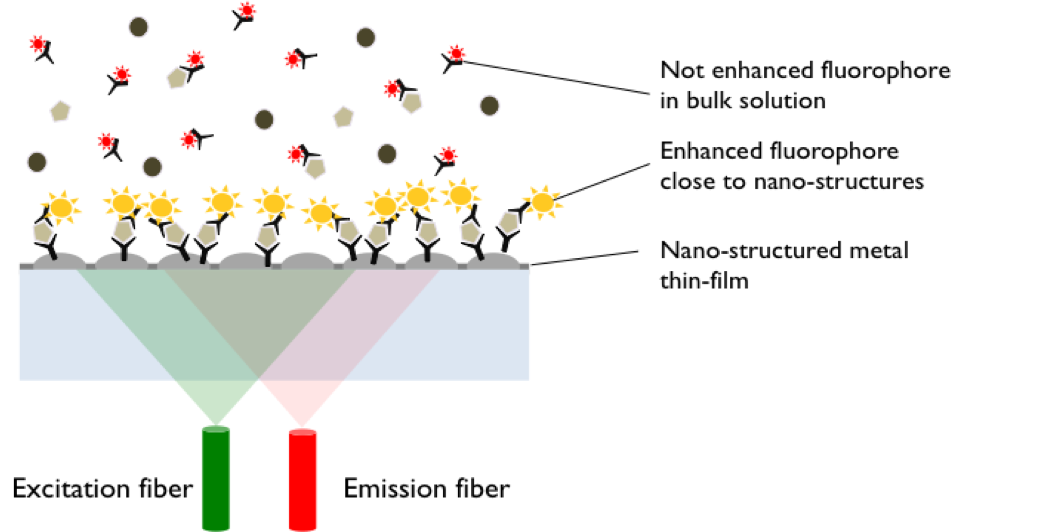
Method: Metal Enhanced Direct Sandwich Fluorescence Immunoassay
Size: 1×96 wells
Sensitivity: LOD <0 pmol/l + 3 SD): 10 pmol/l; LLOQ: 25 pmol/l
Dynamic Range: 0-200 pmol/l
Incubation Time: Overnight
Sample Type: Serum
Sample Size: 10 µl
For Research Use Only
This platform is fully compatible to standard laboratory methodology using 96 well microtiter plate format and assays based on this technology can be run on any standard fluorescence microplate reader.
This assay is regularly delivered standard with AlexaFluor680 labeled detection-antibody. If you want to use a different dye, the following options are available:
- FITC labeled (FIA-1702-F)
- Cy3 labeled (FIA-1702-C3)
- Cy5 labeled (FIA-1702-C5)
Conversion factor: 1 pg/ml = 0.018 pmol/l (MW: 55,7 kD / Monomer)
Assay Background
Asporin, also known as periodontal ligament-associated protein 1 (PLAP1) is a dimeric secreted extracellular matrix protein, which belongs to the small leucine-rich proteoglycan (SLRP) family. It consist out of 380 amino acids and has a highly conserved pro peptide sequence which contains a series of leucine rich repeats and are flanked by two cysteine residues in the C Terminal region. Further it has four cysteine residues that form disulphide bonds as well as aspartic acid repeats in the N-Terminal region.
High levels of Asporin can be found in aorta, uterus and osteoarthritic articular cartilage. Further, moderate levels of aspirin expression can be found in small intestine, heart liver, bladder, ovary, stomach, the adrenal-, thyroid-, and mammary gland. Lower levels of expression can be seen in the trachea, bone marrow and lung. Asporin is known to negatively regulate PDL differentiation and mineralisation as well as it inhibits BMP- dependent activation of SMAD proteins. Further, it directly binds to TGFb-1 subsequently, binds to collagen by way of its LRR domain. Through its interaction with TGFb-1, Asporin negatively regulates chondrogenesis in the articular cartilage by blocking the TGF-beta/receptor interaction on the cell surface and inhibiting canonical TGF-beta/Smad signalling. Moreover, it has the ability to bind calcium, giving it regulatory properties in osteoblast driven mineralisation and regulates FGF2 through direct and indirect interactions. Next to its regulatory properties in terms of cartilage and bone homeostasis, Asporin expression has also been linked to cancer invasion and progression. However, its value as biomarker remains to be established yet.
ASPORIN has been linked to:
- Osteoarthritis
- Lumbar Disc Degeneration
- Tumor Progression
Related Products
FluoBolt-Klotho Fluorescence Immunoassay
FluoBolt-Periostin Fluorescence Immunoassay
FluoBolt Noggin Fluorescence Immunoassay