Anti-Human Antibody (HAHA) ELISA Assay Kit
Anti-Human Antibody ELISA Assay Kit Developed and Manufactured in the USA
Size: 1×96 wells
Sensitivity: 0.1 µg/mL
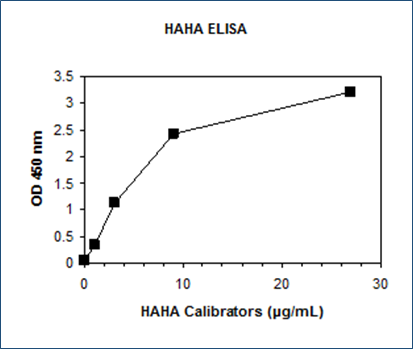
Dynamic Range: 0 – 27 µg/mL
Incubation Time: 2 hours
Sample Type: Serum, Plasma
Sample Size: 50 µL
Alternative Names: Human Anti-Human Antibody ELISA, HAHA ELISA Assay Kit
For Research Use Only
Controls Included
Assay Principle
The Anti-Human Antibody ELISA Assay is designed, developed and produced for the quantitative measurement of HAHA in serum and plasma samples. The assay utilizes the two-site “sandwich” technique with two selected antibodies that bind to HAHA.
The HAHA ELISA assay standards, controls and patient samples are directly added to wells of a microplate that is coated with highly purified human IgG. After the first incubation period, the HAHA binds to the human IgG on the wall of microtiter well and unbound proteins in each microtiter well are washed away. Then a horseradish peroxidase (HRP)-labeled human antibody is added to each microtiter well and a “sandwich” of “well-coated human IgG – HAHA – HRP-conjugated human antibody” is formed. The unbound HRP-conjugated human antibody is removed in the subsequent washing step. For the detection of this immunocomplex, the well is then incubated with a substrate solution in a timed reaction and then measured in a spectrophotometric microplate reader. The enzymatic activity of the immunocomplex bound to HAHA on the wall of the microtiter well is directly proportional to the amount of HAHA in the sample. A standard curve is generated by plotting the absorbance versus the respective HAHA concentration for each standard on point-to-point or 4 parameter curve fit. The concentration of HAHA in test samples is determined directly from this standard curve.
Products Related to the Human Anti-Human Antibody ELISA Assay Kit
Human Anti-IgE Antibody ELISA Assay Kit
Human Anti-Mouse Antibody (HAMA) ELISA
Mouse Anti-Human Antibody (MAHA) ELISA Assay Kit