
What is Actiflash intended for?
Actiflash is used to convert your inducible ERT model into a photo-inducible one. Use it if you want to control transcription (using Gal4-UAS) or induce recombination (using Cre-lox) in space and/or time for in-vivo cell tracking experiments and more. Actiflash is for research use only
Features
-
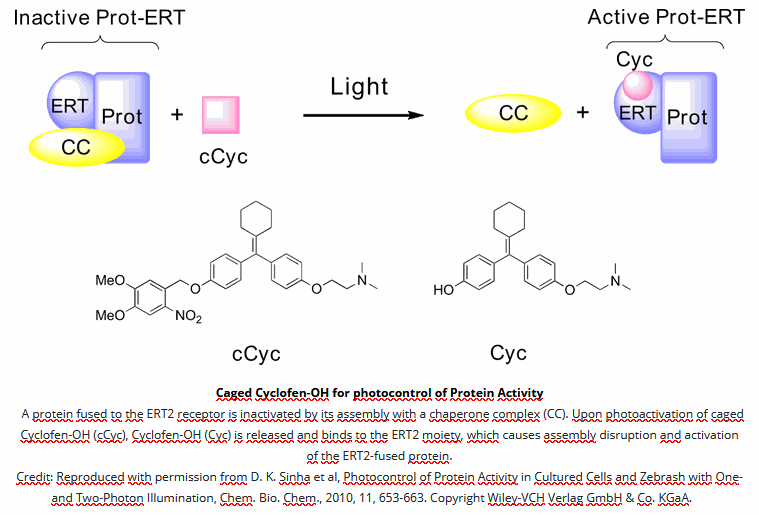
- Wide applicative scope – Technology capitalizing on the versatile use of Tamoxifen-OH for controlling functions of multiple types of proteins.
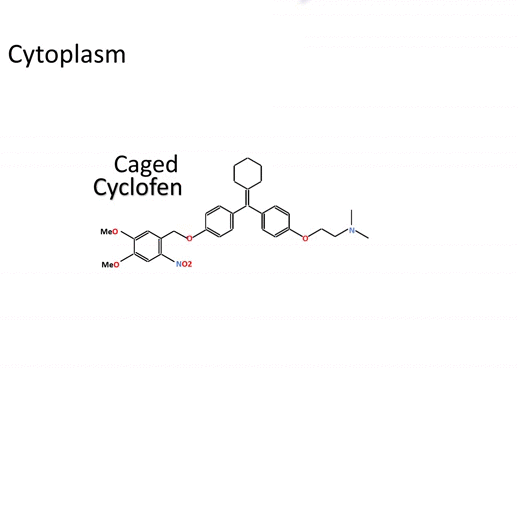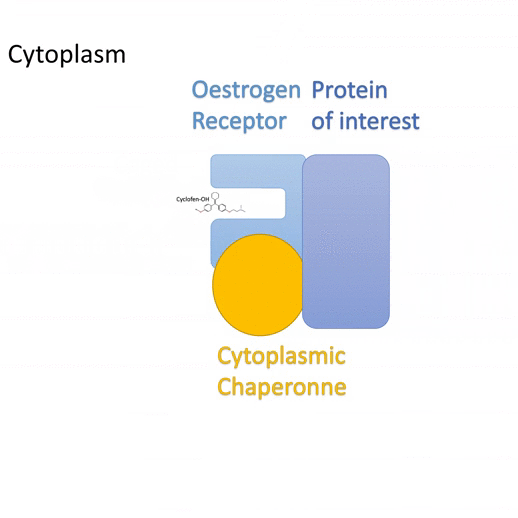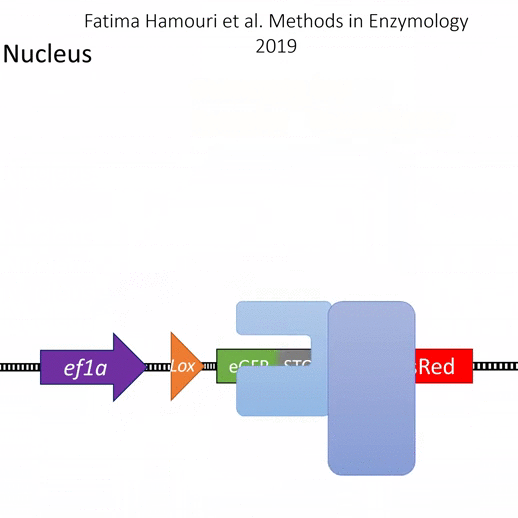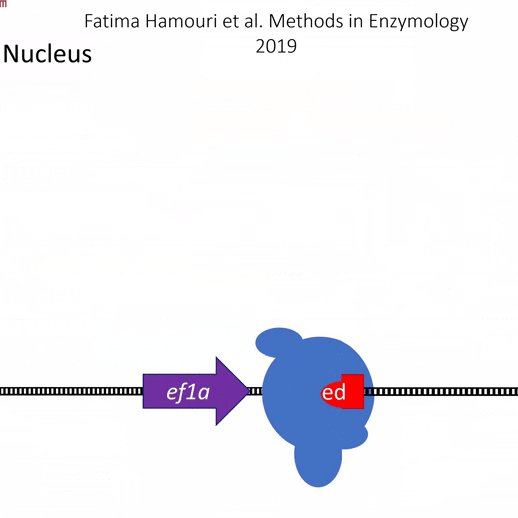
- Simple conditioning – Caged Cyclofen-OH is cell-permeant and can be added either in the external medium or directly injected for conditioning.
- Excellent chemical stability – Caged Cyclofen-OH does not generate any basal activation of protein function and it benefits from an excellent temporal resolution upon uncaging.
- Favorable wavelength ranges for uncaging – Uncaging requires either UV-A light or a strong IR laser. Visible light is inactive, which facilitates the experiments with biological samples.
- Photochemical stability – Caged Cyclofen-OH liberates Cyclofen-OH, which is photostable in contrast to Tamoxifen-OH encountering photodegradation under illumination.
Method of Use:
Calibration of the Actiflash concentration
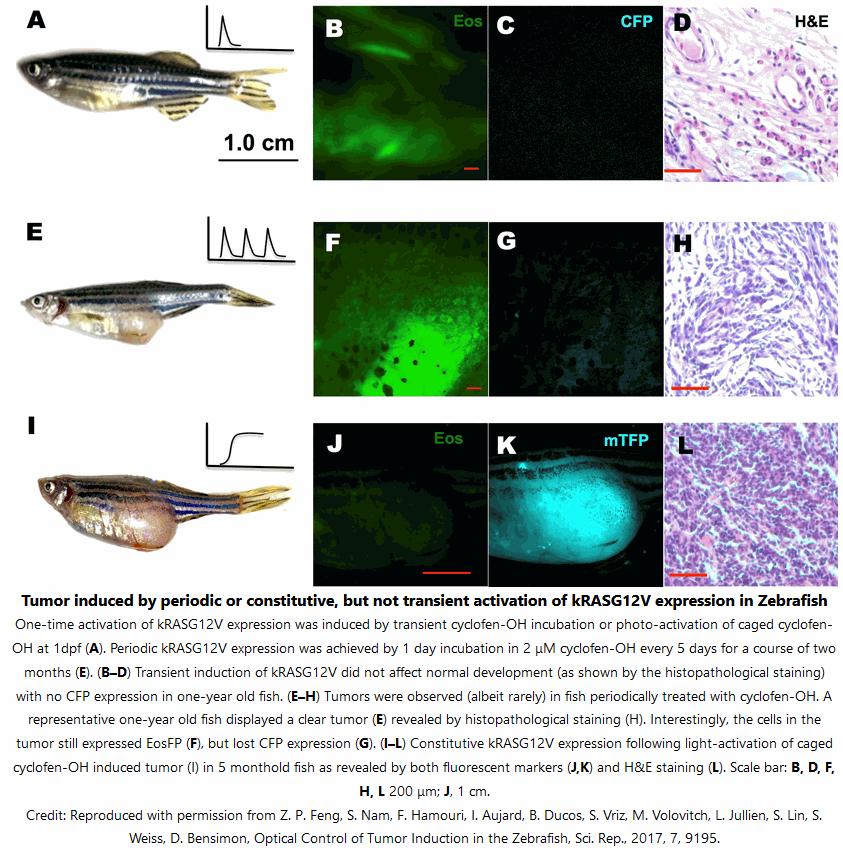
It is advised to first establish the extent of phenotype sought for as a function of the Tamoxifen-OH concentration. Then the concentration of Actiflash used for sample conditioning is fixed at Tamoxifen-OH concentration causing 100% of the desired phenotype (in general 3-5 μM in cultured cells and zebrafish embryos).
Conditioning protocol
Incubate your samples in a serum-free medium for 90 mins, away from light.
Photoactivation
Illumination of Actiflash may be performed with UV (325-425nm range) light or multiphoton excitation (at 750 and 1064 nm with two- and three-photon excitation, respectively) to release Cyclofen-OH. You can use either benchtop UV lamps or light sources installed on microscopes.
The calibration of the photoactivation
The objective is to provide enough photons to exhaust the conversion of the Actiflash but without generating detrimental side-effects on the biological sample. Simply analyze the phenotype recovery with decreasing illumination duration. Then determine the shortest illumination duration leading to 100% uncaging of Actiflash.
5 Key Conditions of Success
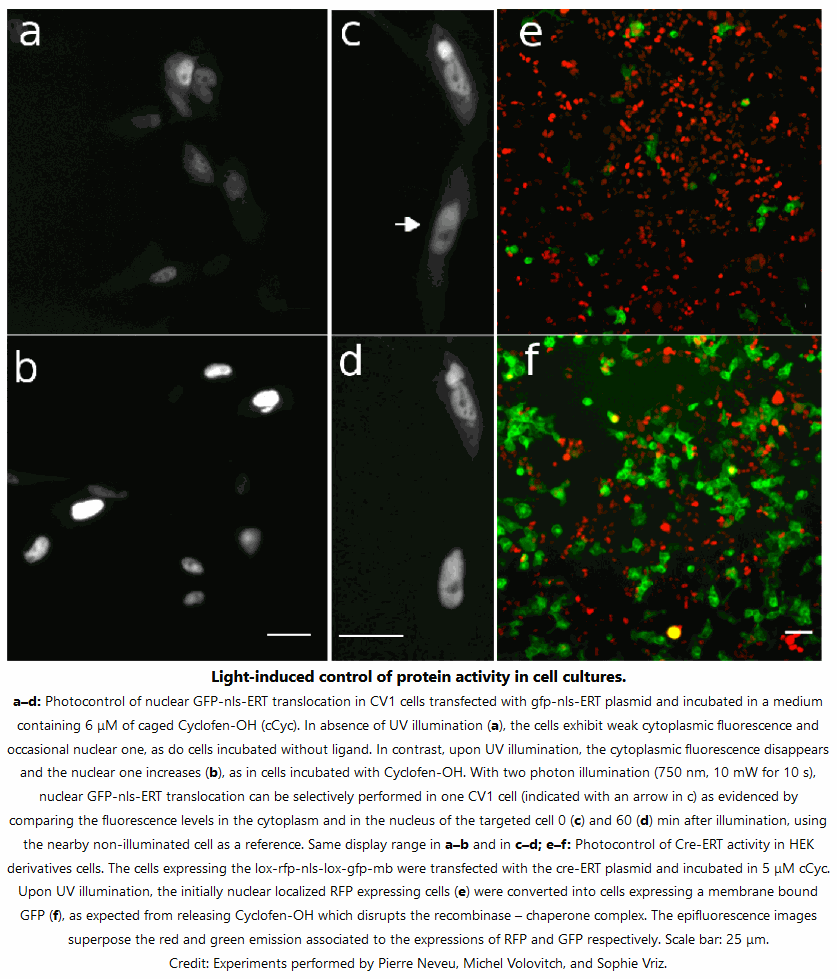
- Actiflash is provided as a powder. Resuspend it with DMSO (warning: no water).
- Only unfreeze the aliquots you need for your experiment.
- Carefully calibrate the photoactivation of Actiflash (concentration for conditioning, power of the light source, geometry and duration of illumination). Then always work under the same conditions.
- Use a serum-free medium during the incubation step of the cell lines.
- In the dark, just before photoactivation, wash your samples (unnecessary for two-photon laser experiments).
For more information about this product or any others from the Microscopy line, contact us here.
