What
is ANP?
The first
natriuretic peptide was identified in 1983 and named atrial natriuretic peptide
(ANP). ANP is a 28-amino acid polypeptide resulting from the C-terminal end of
the prohormone proANP. It is largely produced in the cardiac atria, and ANP is
quickly secreted in response to atrial stretching. ANP has the following
physiological effects:
- Increases glomerular
filtration rate by dilating afferent arterioles - Inhibits the collecting ducts from reabsorbing sodium,
both directly and indirectly (by inhibiting aldosterone secretion) - Inhibits release of renin
The renin-angiotensin system and ANP
function antagonistically in the maintenance of fluid/electrolyte balance and
blood pressure.
Normal hearts
secrete extremely small amounts of ANP, but elevated levels are found in
patients with left ventricular (LV) hypertrophy and mitral valve disease.
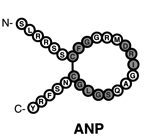
Biochemical structure of ANP:
ANP is synthesized as a 126 amino acidprecursor protein which is cleaved to produce a
96 aminoacid amino-terminal fragment and a 28 amino acid
carboxyl-terminal fragment. The carboxyl-terminal 28 amino acid fragment is the biologically active peptide and
has shorter half-life than the amino-terminal fragment.
Conserved residues are shaded and the line
within the ring represents the disulfide bond.
What
is proANP?
ProANP is the prohormone of ANP (ANP is stored as a
126–amino acid prohormone (proANP1–126)). After this prohormone is released it is then cleaved into equimolar amounts of the biologically active C-terminal peptide (ANP (99–126)) and the residual N-terminal peptide (NT-proANP (1–98)) in response to atrial wall stretch.
α-ANP has a half-life of 3-4 minutes thus is quickly cleared from circulation. ProANP (1-98) on the other hand, has a significantly longer half-life of 60-120 minutes leading to much higher concentrations in blood as compared to α-ANP.
Why Measure proANP (1-98)?
Atrial natriuretic peptide (ANP) has an important physiological roles in fluid homeostasis and cardiac pathology. The longer half life of proANP (1-98) offers advantages for measuring the ANP in the blood over α-ANP due to its lack of sensitivity to the pulsatile secretion of ANP. In fact,proANP (1-98) may provide better insight to the ANP status and its chronic secretion in the blood. Studies have shown that proANP serves as a
valuable and stable marker for several areas of research ranging from sepsis to
predicting and aiding in risk stratification for heart failure.
Diseases/conditions associated with increased ANP levels:
- Diabetes and Obesity
- Renal Disease/Renal dysfunction
- Hypertension
- Pneumonia
- Asthma attacks
- Recent heart attack
- Heart failure/Cardiac impairment
- Cardiomyopathy
- Sepsis
- Sleep apnea
References:
- Haviv, M. et al. “Atrial Natriuretic Peptidein Children with Pneumonia.” Pediatric Pulmonology 40:306–309(2005).
- Hoffman et al. “Prognostic Value of Plasma N-Terminal Pro-Brain Natriuretic Peptide in Patients With Severe Sepsis.” Circulation; 2005:112:527-534.
- Lauridsen, Bo K et al. “ProANP Plasma Measurement Predicts All-Cause Mortality in Acutely Hospitalized Patients: A Cohort Study.” BMJ Open 2013;3:e003288 doi:10.1136/bmjopen-2013-003288.
- Moro, Cedric et al. “Natriuretic Peptides: New Players in Energy Homeostasis.” Diabetes 2009; 12: 2726-2728.
- Robichaud, A. et al. “Plasma Atrial Natriuretic Peptide During Spontaneous Bronchoconstriction in Asthmatics. Peptides 1995;16(4):653-6.
- Suzuki, T et al. “The Role of the Natriuretic Peptides in the Cardiovascular System.” Cardiovascular Research 2001; 51: 489–494.
Related Resources Citing EagleBio Kits:
Related Kits:
proANP ELISA Assay Kit
Cardiovascular Assay Kits
Related News:
EagleBio Spotlight: Natriuretic Peptides
EagleBio Spotlight: BNP
EagleBio Spotlight: CNP
Interesting Study Highlighting Eagle Biosciences’ BNP Fragment ELISA Assay Kit