Stencell
Stencell was developed and manufactured in France by Idylle Labs
What is Stencell intended for?
Stencell is used notably for wound-healing assay & immunocytochemistry experiments. It is very helpful when you are working with super expensive reagents or very rare cell lines, and when you want to test various experimental hypothesis.
Stencell for wound-healing experiments
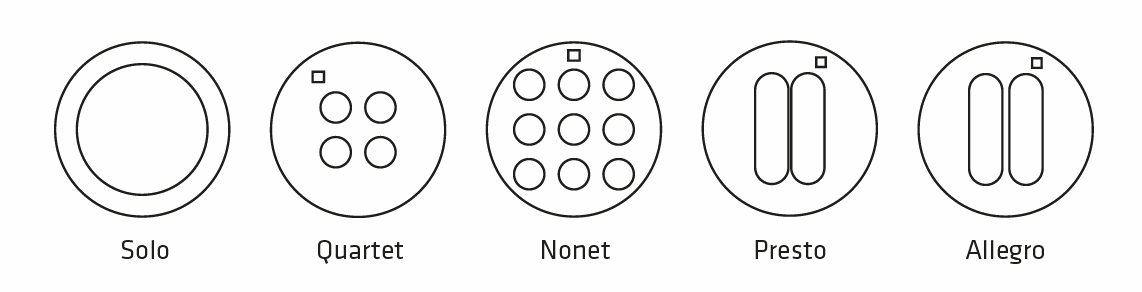
The Designs
Each order is composed of 2 sheets of 50 identical stencils (=100 stencils). Upon receipt of your purchase order, you will be able to choose either the same design for your 2 sheets, or 2 different designs for your 2 sheets (1 per sheet of 50).
-
- Solo: 1 circular well – Diam. 12 mm
- Quartet: 4 circular wells – Diam. 3 mm
- Nonet: 9 circular wells – Diam. 3 mm
- Presto: 2 oblong wells spaced by 0.35 mm
- Allegro: 2 oblong wells spaced by 0.62 mm
Features
- Design your PDMS – Our design process offers total freedom to turn your idea into a PDMS.
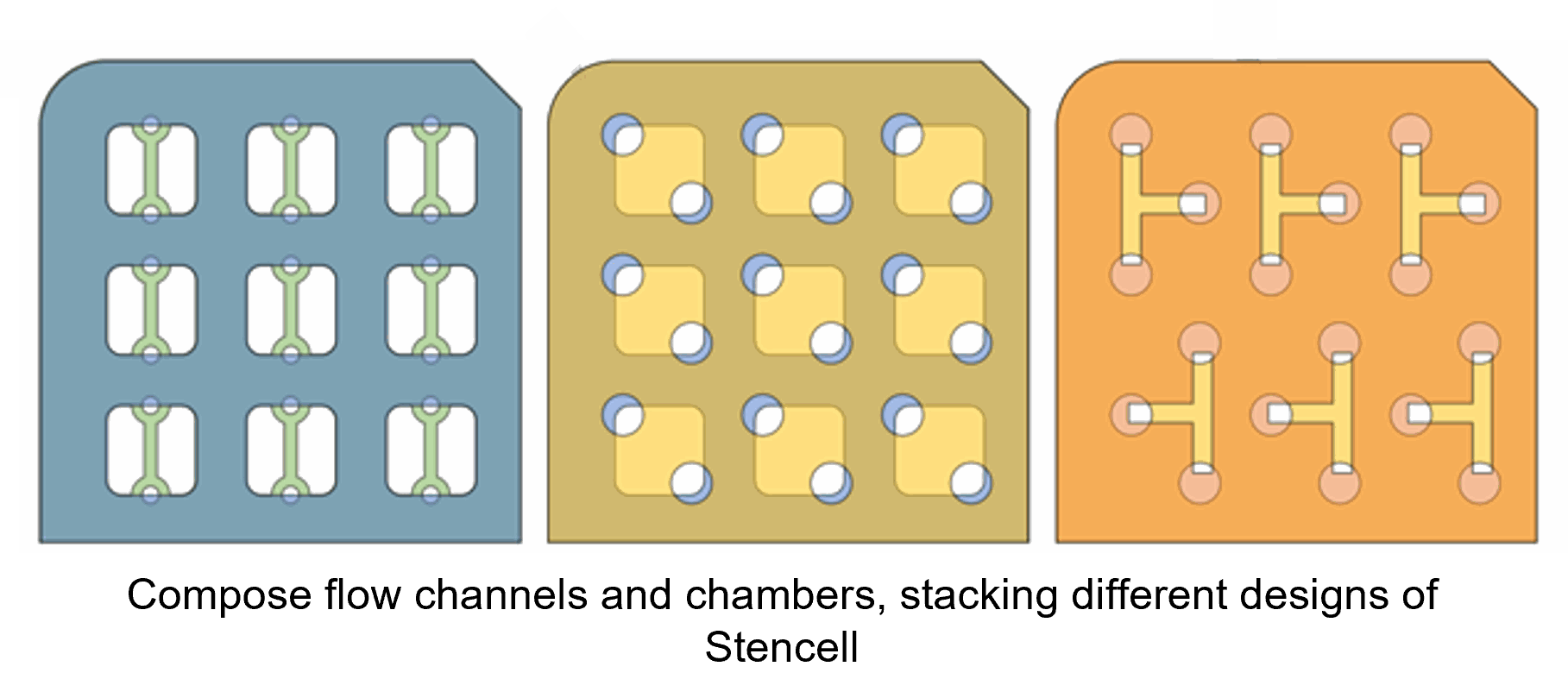
- Stack and Play – Compose flow channels and chambers, stacking different designs of Stencell.
- Stick it to a variety of culture labware – So far, it has been successfully stuck onto the following: glass slides, plastic dishes, Transwell(R) inserts, Polyacrylamide gels.
- Remove it – Stencell is not glued. It can then be removed to trigger cell migration, switch from flow chamber to open window. (Very useful for wound healing experiments)
- It is compatible with imaging – These thin sheets of silicone are fully transparent, without autofluorescence.
- Parallelized and standardized experiments – One multiwell Stencell can perform several experimental conditions.
- It saves samples and reagents – Only a few microliters are required.
Key Conditions of Success
- When you remove the inner elements of your Stencell, use 2 tweezers: 1 to remove and the other 1 to press close to the junction in order to avoid breaking the Stencell. Alternatively, you can cut the junctions with a scalpel.
- When you put the Stencell on top of your culture substrate, softly remove the bubbles and stick the Stencell, patting it with tweezers.
- Depending on the substrate hydrophobicity, the droplet might not completely fill the well. You can:
- Either fill the well with the standard volume. Then, help the droplets stick to the Stencell by connecting the liquid next to the Stencell walls using the pipette tip.
- Or, you may add an excess volume of liquid (e.g. 30 μL) and remove it afterwards (e.g. 10 μL).
- But: do not overfill (e.g. > 30 μL) the wells, otherwise two droplets may merge.
- When you put your substrate and cells in the incubator. Wait for 1 to 2 hours until cells spread and fully occupy the windows space. If you need to wait longer, be sure that the droplets do not completely evaporate.
Related Products