Guselkumab is a human monoclonal antibody of the IgG1λ class that specifically targets the p19 subunit of interleukin-23 (IL-23), a pro-inflammatory cytokine integral to the CD4+ T-helper 17 (Th17) cell-mediated immune response. This cytokine pathway is crucial in the pathogenesis of psoriatic plaque formation, making IL-23 a key biomarker in both clinical and research contexts for inflammatory skin disorders such as psoriasis. Guselkumab’s selectivity for IL-23p19 allows it to block the cytokine’s interaction with its receptor on keratinocytes and dendritic cells, inhibiting downstream production of additional inflammatory mediators and chemokines.
Clinically, guselkumab is indicated for patients with chronic inflammatory conditions characterized by IL-23 and IL-12 overexpression, particularly moderate-to-severe plaque psoriasis and potentially other autoimmune skin diseases. By targeting IL-23p19, guselkumab reduces keratinocyte hyperproliferation and the formation of psoriatic plaques, leading to significant improvements in skin clearance and symptom relief. These effects are measurable through decreased expression of downstream biomarkers such as IL-6, IL-8, and various matrix metalloproteinases in patient samples.
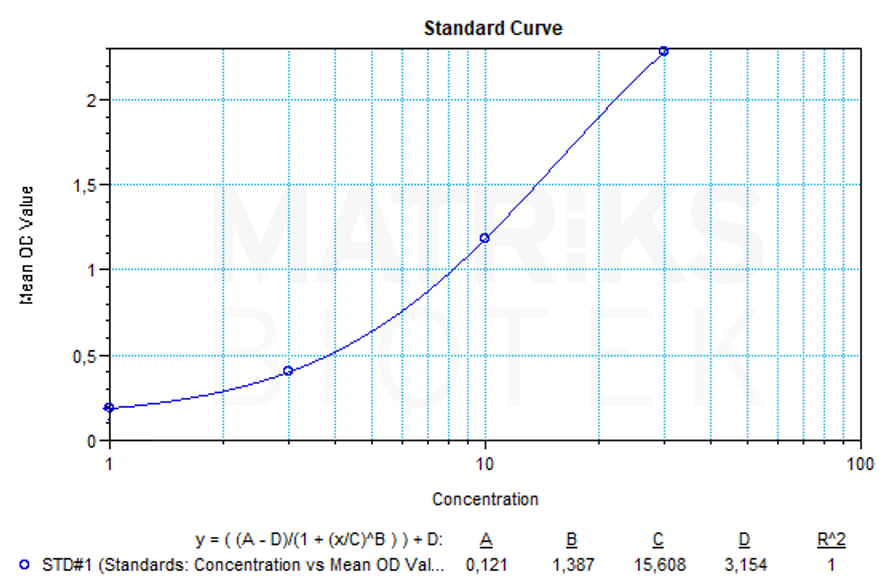
In research settings, guselkumab serves as a valuable biologic for understanding the role of the IL-23/Th17 axis in autoimmune pathology. It also provides insight into therapeutic mechanisms, drug development, and immunogenicity profiling. As with other biologics, therapeutic drug monitoring (TDM) and antidrug antibody (ADA) analysis are important in evaluating treatment efficacy, pharmacokinetics, and immune response variability in different patient populations.
This product is manufactured in Turkey by Matriks Biotek.