Etanercept is a dimeric fusion protein consisting of the extracellular ligand-binding domain of the human 75 kDa tumor necrosis factor receptor (p75 TNFR) fused to the Fc portion of human IgG1. It is produced using recombinant DNA technology in Chinese hamster ovary (CHO) cell lines and is composed of 934 amino acids. Its design allows it to effectively neutralize tumor necrosis factor (TNF) by binding to soluble TNF molecules, preventing their interaction with cell surface TNF receptors and thereby modulating inflammatory responses.
In clinical settings, etanercept is used to treat autoimmune diseases characterized by excessive TNF activity, such as rheumatoid arthritis, psoriasis, and ankylosing spondylitis. By targeting and neutralizing TNF, etanercept reduces the expression of adhesion molecules, cytokines, and enzymes like matrix metalloproteinase-3 (stromelysin), thereby lowering inflammation and halting tissue damage. Its biological activity depends on binding to either p55 or p75 TNF receptors.
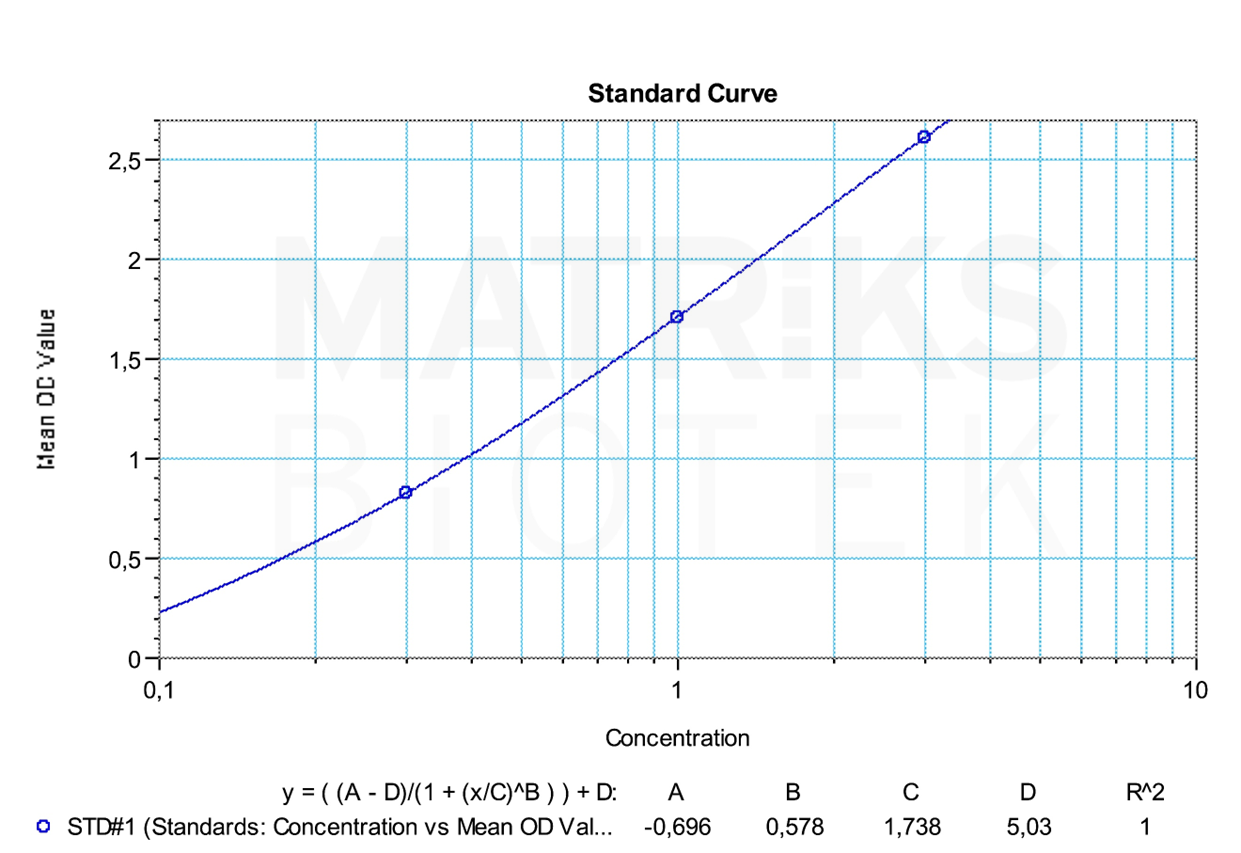
For research and therapeutic drug monitoring (TDM), etanercept’s pharmacokinetics, patient compliance, and immunogenicity profile must be closely observed. As with other biologics, biosimilar versions exist, but due to molecular complexity and variable manufacturing processes, they may differ immunologically. Monitoring the formation of anti-drug antibodies (ADAs) is critical, as these can impact drug efficacy and lead to adverse events, underscoring the importance of precision in both clinical and investigational use of etanercept.
This product is manufactured in Turkey by Matriks Biotek.