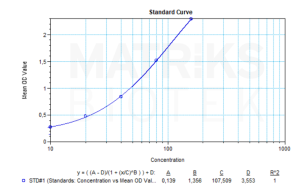
Antibodies to Vedolizumab, including anti-drug antibodies (ADAs), may develop in patients receiving vedolizumab therapy for gastrointestinal inflammatory disorders. These antibodies can potentially bind to the drug, with or without neutralizing its therapeutic effect. Neutralizing antibodies (NAbs) specifically inhibit vedolizumab’s function by preventing its interaction with α4β7 integrin, thereby reducing the drug’s efficacy in blocking lymphocyte trafficking to gut-associated lymphoid tissue. In contrast, non-neutralizing antibodies may not interfere with the mechanism of action but could impact pharmacokinetics or increase the risk of hypersensitivity reactions.
In a research context, quantifying anti-vedolizumab antibodies is critical for understanding immunogenicity profiles and treatment resistance mechanisms. Clinically, therapeutic drug monitoring (TDM) that includes both vedolizumab trough levels and ADA screening helps optimize dosing strategies, identify patients at risk of treatment failure, and support decision-making for therapy adjustments. Monitoring anti-vedolizumab antibodies can therefore enhance patient management and improve long-term outcomes in chronic inflammatory conditions such as ulcerative colitis and Crohn’s disease.
This product is manufactured in Turkey by Matriks Biotek.