Anti-omalizumab antibodies are anti-drug antibodies (ADAs) that develop in response to treatment with omalizumab, a monoclonal antibody targeting immunoglobulin E (IgE). These antibodies can be formed as part of the body’s immune response to the biologic therapy and may bind to omalizumab, potentially impacting its therapeutic effect. While not all anti-drug antibodies are neutralizing, those that interfere with omalizumab’s binding to IgE may reduce the drug’s clinical efficacy and alter pharmacokinetics.
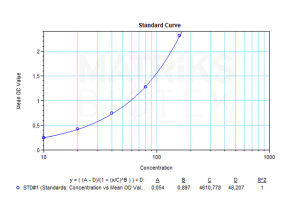
In a research and clinical setting, the detection and monitoring of anti-omalizumab antibodies are critical components of therapeutic drug monitoring (TDM). TDM helps optimize treatment by identifying patients who may be experiencing reduced drug response due to immunogenicity. Monitoring ADAs is also essential in understanding adverse reactions, treatment failure, or the need for dose adjustments.
Regular assessment of anti-omalizumab antibodies allows clinicians to maintain effective treatment strategies for patients with asthma or chronic idiopathic urticaria. This biomarker is especially useful in personalizing biologic therapy and ensuring continued benefit from omalizumab in populations that might otherwise experience variable responses due to immune-mediated interference.
This product is manufactured in Turkey by Matriks Biotek.