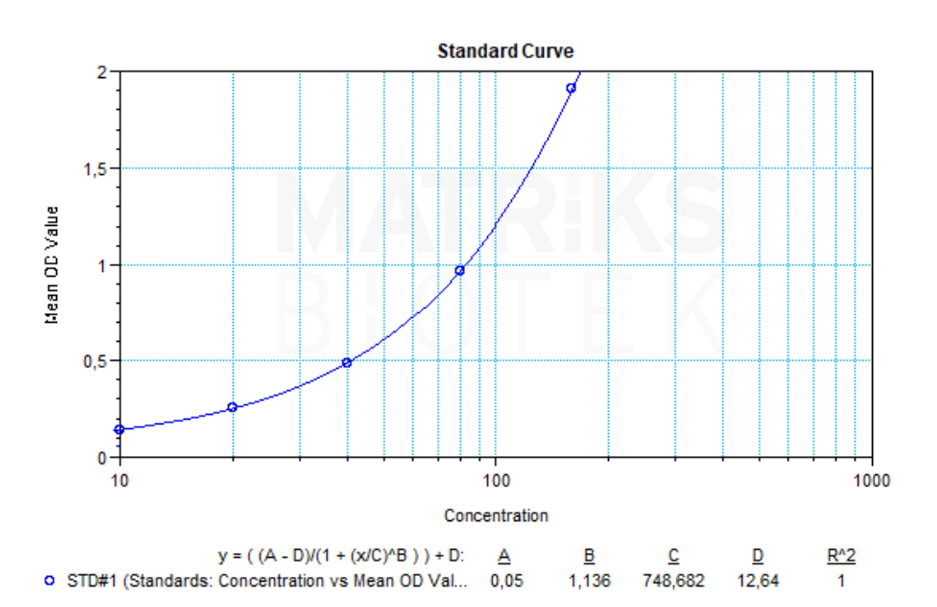
Anti-eculizumab antibodies are immune responses developed against eculizumab, a recombinant humanized monoclonal antibody targeting complement protein C5. Eculizumab prevents the cleavage of C5 to C5a and C5b and the subsequent formation of the terminal complement complex C5b-9. This mechanism plays a critical role in managing complement-mediated diseases by preventing immune system overactivation. The presence of anti-drug antibodies (ADAs) may impact the therapeutic efficacy of eculizumab by altering its pharmacokinetics or inducing loss of clinical effect.
Monitoring anti-eculizumab antibodies in both research and clinical settings is important for assessing treatment response and guiding therapeutic adjustments. These antibodies may influence disease control in patients with paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (aHUS), and neuromyelitis optica spectrum disorder (NMOSD), where eculizumab is used to reduce complement-mediated damage. By identifying the development of ADAs, clinicians can better manage patients with suboptimal responses or adverse reactions.
In research, anti-eculizumab biomarker assays are essential tools for evaluating immunogenicity during clinical trials and for biosimilar development. Understanding ADA prevalence and its clinical consequences supports safer biologic use and the development of optimized treatment regimens. Given the potential for serious infections due to complement inhibition, biomarker tracking may also help stratify risk and personalize long-term therapy.
This product is manufactured in Turkey by Matriks Biotek.