Humanized Anti-Her2/neu (Herceptin / Trastuzumab) ELISA Kit
Trastuzumab (Herceptin) is a humanized monoclonal antibody that targets the human epidermal growth factor receptor 2 (HER2), a transmembrane tyrosine kinase receptor involved in cell proliferation and survival. HER2 is overexpressed in approximately 15–20% of breast cancers and in a subset of gastric and gastroesophageal cancers, often correlating with aggressive disease and poor prognosis. Trastuzumab binds to the extracellular domain of the HER2 receptor, inhibiting its signaling, promoting antibody-dependent cellular cytotoxicity (ADCC), and preventing receptor dimerization. This mechanism effectively halts tumor growth and facilitates immune-mediated destruction of cancer cells.
In clinical settings, Trastuzumab is used primarily in the treatment of HER2-positive breast cancer, both in early-stage and metastatic disease. It is often combined with chemotherapy agents such as paclitaxel or docetaxel, and its use has significantly improved survival outcomes for patients with HER2-positive tumors. Beyond breast cancer, Trastuzumab is approved for HER2-positive metastatic gastric and gastroesophageal junction cancers. In research, it serves as a model therapeutic for studying monoclonal antibody mechanisms, resistance pathways (e.g., HER2 mutations or compensatory signaling), and the development of biosimilars. It is also a key component in evaluating novel HER2-targeted therapies, including antibody-drug conjugates and bispecific antibodies.
This Humanized Anti-Her2/neu (Herceptin / Trastuzumab) ELISA Kit is manufactured in USA by Eagle Biosciences.
| Size | |
| Sensitivity | 0.0077 µg/mL |
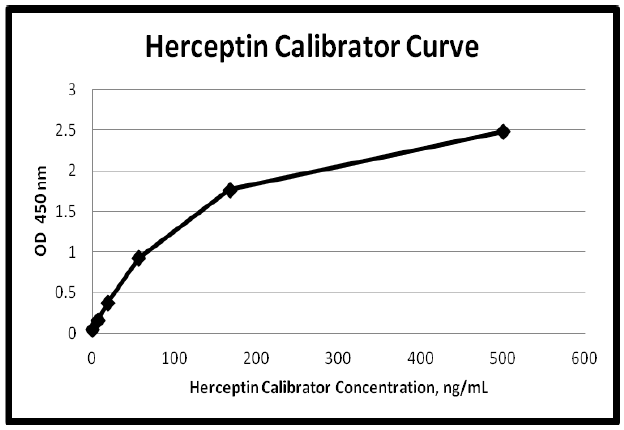
| Dynamic Range | 0.245 – 500 ng/mL |
| Incubation Time | 2 hours |
| Sample Type | Serum, EDTA plasma |
| Storage | 2-8°C |
| Alternative Names | Trastuzumab, Trazimera, Anti-HER2 antibody |