Anti-Golimumab antibodies refer to the antidrug antibodies (ADAs) generated in response to treatment with Golimumab, a human IgG1κ monoclonal antibody that targets tumor necrosis factor-alpha (TNFα). Golimumab functions by binding and inhibiting both soluble and transmembrane forms of TNFα, thereby suppressing downstream inflammatory responses. However, like other biologics, Golimumab can elicit an immune response in some patients, leading to the production of ADAs that may neutralize its therapeutic effect or alter its pharmacokinetics.
In a research and clinical setting, detection of Anti-Golimumab antibodies serves as a biomarker for immunogenicity, treatment monitoring, and potential loss of efficacy. The presence of these antibodies may correlate with increased TNFα activity, elevated inflammatory markers (such as IL-6, IL-8, CRP), or clinical signs of treatment failure in chronic inflammatory conditions. Monitoring Anti-Golimumab levels can also help optimize dosage, identify the need for therapy modification, and predict the risk of adverse immune-mediated reactions.
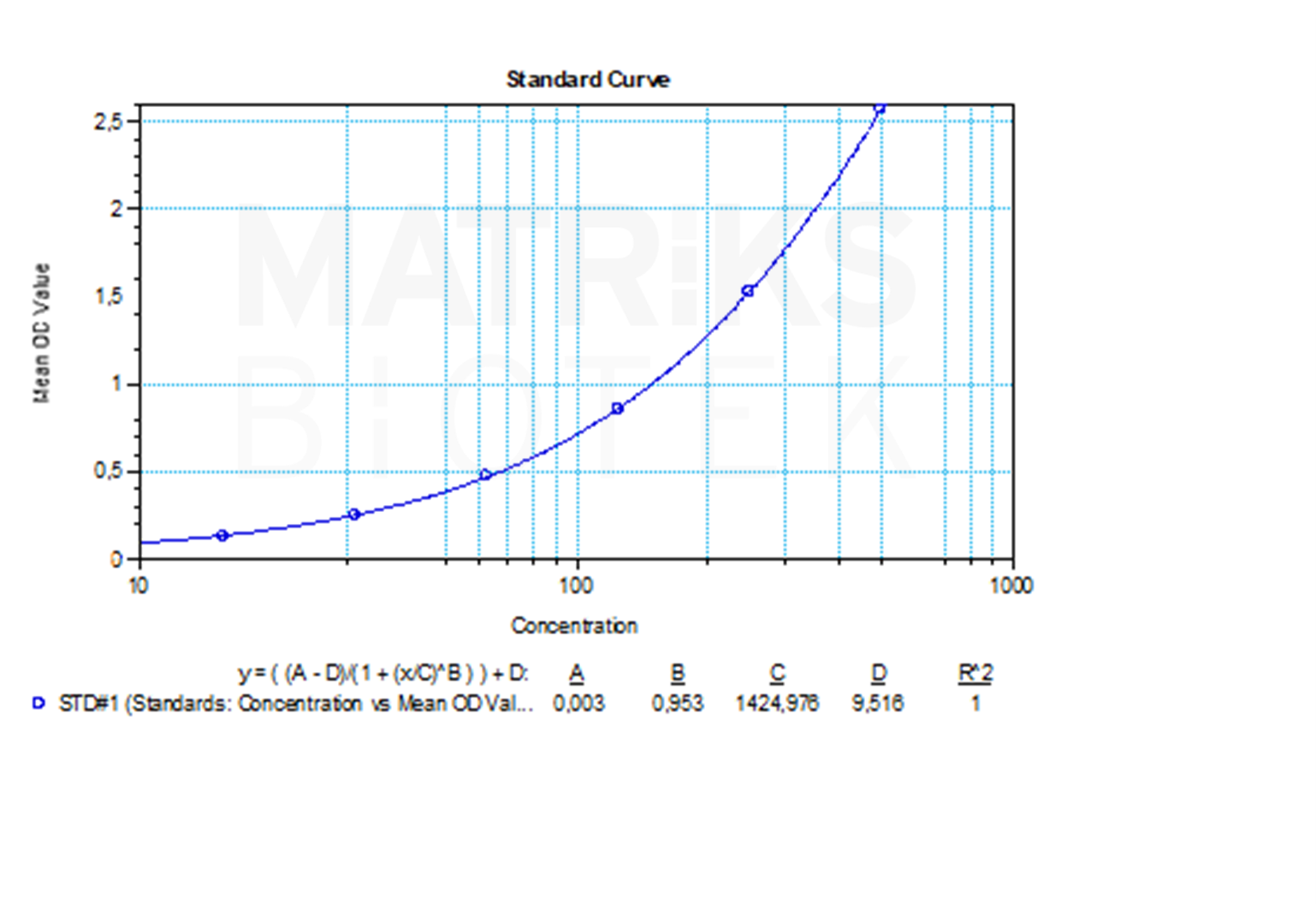
Therapeutic drug monitoring (TDM) is crucial in this context to assess both drug and ADA levels. The integration of Anti-Golimumab biomarker analysis into TDM protocols enables personalized treatment plans, helps avoid unnecessary drug escalation, and ensures patients maintain therapeutic benefit while minimizing risks associated with biologic therapies.
This product is manufactured in Turkey by Matriks Biotek.