Pertuzumab is a recombinant humanized monoclonal antibody that targets the extracellular dimerization domain (subdomain II) of the human epidermal growth factor receptor 2 (HER2) protein. By binding to this domain, pertuzumab prevents the HER2 receptor from pairing (dimerizing) with other HER family members, thereby inhibiting the downstream signaling pathways responsible for tumor growth and proliferation. This mechanism makes pertuzumab particularly effective in HER2-positive cancers, where the HER2 protein is overexpressed and contributes to aggressive tumor behavior.
Clinically, pertuzumab is approved for the treatment of adults with HER2-positive metastatic breast cancer. It is especially used in cases where the cancer has either spread beyond the breast or recurred locally and is not amenable to surgical removal. Pertuzumab is typically administered in combination with trastuzumab and chemotherapy agents like docetaxel to enhance its therapeutic effect by targeting HER2 through complementary mechanisms.
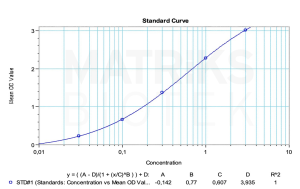
In the research setting, pertuzumab serves as a critical tool in studying HER2-driven oncogenesis and therapeutic resistance. It is also a candidate for therapeutic drug monitoring (TDM) in clinical practice to ensure optimal drug levels, minimize toxicity, and evaluate immunogenicity. The development of biosimilars and anti-drug antibodies (ADAs) to pertuzumab further highlights its significance in biologic drug research and underscores the importance of precise analytical tools for pharmacokinetics and immune response profiling in personalized oncology treatment.
This product is manufactured in Turkey by Matriks Biotek.